Back to Basics – The Ozone Hole
J. D. Shanklin
(British Antarctic Survey)
Ozone
is one of the key radiatively active gases in the stratosphere. Where there is less ozone the stratosphere tends
to be colder and at any one location there is a good correlation between the
100 or 70 hPa temperature and the total column ozone. Ozone absorbs solar ultraviolet light and where there is less
ozone more ultraviolet will reach the surface, posing risks to human health.
Ozone is a compound of oxygen containing three atoms instead of two as in the oxygen gas that sustains life. It was discovered in 1839 by a Swiss chemist, Christian Friedrich Schonbein. In high concentration ozone is a bluish green gas, with very strong oxidising properties. It is a toxic, irritating gas, often encountered in surface air pollution episodes, when it can trigger asthma and irritate mucous membranes. Dry air consists of 78% nitrogen and 21% oxygen and normally trace amounts of other gases, principally argon, water and carbon dioxide are present. The concentration of ozone is usually only a few parts per million and even in the ozone layer it is only one part in 100,000.
Ozone concentrations at the surface were first measured reliably by Robert Strutt (later 4th Lord Rayleigh) in 1918 using spectra of a hydrogen lamp recorded through five kilometres of air. These measurements showed that ozone concentration could not be uniform throughout the atmosphere as a higher concentration was required to explain the sharp cut off at around 300 nanometres (nm) seen in stellar spectra. Three years later Fabry and Buisson used spectrographic techniques to demonstrate that its principal atmospheric location is in the stratosphere, though it was not until the 1930s that the actual vertical distribution was first measured. It was soon recognised that measuring the variation in the total ozone column was of meteorological interest and Professor G M B Dobson developed a prototype ozone spectrophotometer in the 1920s. His instrument is still the standard today and around 120 Dobson spectrophotometers have been built.
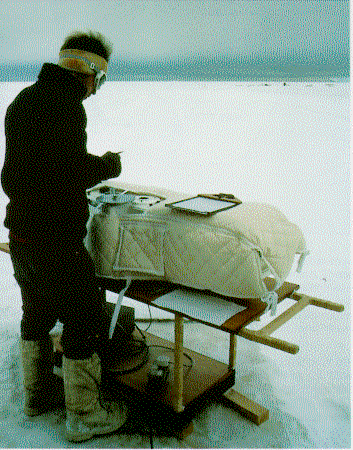
Figure 1 – The Dobson ozone
spectrophotometer at Halley station, during an intercomparison of instruments.
[British Antarctic Survey]
Dobson’s instrument measures ozone by comparing the intensities of two wavelengths of light from the Sun, one of which is absorbed quite strongly by ozone, whilst the other is only weakly absorbed. The ratio of the intensities varies with the amount of ozone present in the atmosphere, and a well-calibrated instrument can measure ozone amounts to within a few percent. The instrument uses wavelengths between 305 and 340 nm and these are selected by means of prisms and a series of slits. It was initially a photographic instrument, but photocells were introduced in the mid 1930s and a photomultiplier in 1946.
The Sun emits radiation in all parts of the electromagnetic spectrum, roughly 7% in the ultraviolet between 200 and 400 nm, 41% in the visible between 400 and 760 nm and 52% in the infra red. The ultraviolet part of the spectrum is further divided into UV-A, UV-B and UV-C. UV-A lies between 315 and 400 nm and gives rise to a suntan and ageing of the skin. UV-B lies between 280 and 315 nm and is the damaging part of the spectrum. UV-C, which is totally absorbed by the atmosphere before it can reach the ground, lies between 200 and 280 nm.
Ozone is created by the photochemical destruction of oxygen, which liberates a free oxygen atom and this can then combine with an oxygen molecule to create ozone. A third molecule is required to take away excess energy. The dissociation requires ultraviolet light of wavelength shorter than 240 nm. Sidney Chapman gave the first description of the photochemistry in 1930.
O2 + hf -> 2O
O2 + O + M -> O3 + M
Ozone can be dissociated by light of wavelength shorter than 1100 nm:
O3 + hf -> O2 + O
The free oxygen atom thus created quickly finds another oxygen molecule and the ozone is reformed with the net result of absorbing the solar radiation and inputting the energy into the atmosphere as thermal energy. The process is very efficient and virtually all radiation between 200 and 310 nm is absorbed, despite the relatively low concentration of ozone. The main ozone absorption bands in the ultraviolet are the Hartley (around 200 – 300 nm) and Huggins (around 300 – 350 nm), and there is the weak Chappuis band in the visible (440 – 740 nm). In the lower stratosphere, below about 30 km, ozone has a long lifetime, and the ozone mixing ratio can be used to trace atmospheric motions.
A typical value for the total amount of ozone in a vertical column of our atmosphere is around 300 Dobson Units (DU), or 300 milli-atmosphere-centimetres, which corresponds to a layer of ozone 3 mm thick at the Earth's surface. This 3 mm is in reality spread through the column, with the bulk of it lying between the tropopause, at 10 to 12 km altitude, and 40 km, with a maximum at around 17 to 25 km altitude depending on location.
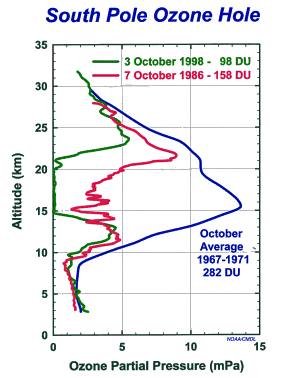
Figure 2 – South pole
ozonesonde profiles, showing the progressive thinning of the ozone layer during
October. [NOAA/CMDL]
Early studies of the ozone layer suggested a good correlation between changes in the total ozone column and synoptic scale weather systems. The prospect of being able to use this in weather forecasting encouraged the setting up of a global network of ozone monitoring stations, however this was a blind alley. Developments in computer power have led to the use of numerical modelling as the key tool in weather prediction.
Day to day changes in ozone concentration are
governed by motions in the stratosphere.
These movements are a combination of horizontal and vertical
transport. The ozone creation region
above 50 km acts as a source or sink, so that descent of air acts to increase
the total ozone column, whilst ascent of air acts to reduce it. Sustained ascent can lead to significant
apparent ozone depletion and is responsible for some of the so-called mini
ozone holes.
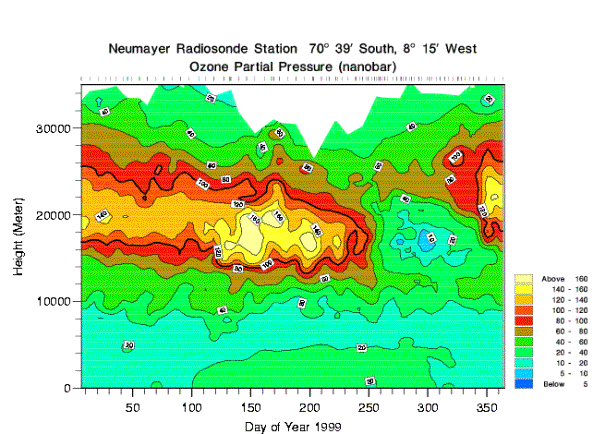 Figure
3 - The annual cycle of ozone in 1999 at Neumayer. [Alfred Wegener Institute]
Figure
3 - The annual cycle of ozone in 1999 at Neumayer. [Alfred Wegener Institute]
The longest running ozone data set in the world
is that from Arosa in Switzerland, and this goes back to 1926. It shows a downward trend of a few percent
per decade since 1970, with the depletion mostly occurring during the winter
and spring. The Antarctic record is
shorter still and in common with most Antarctic data sets it begins in 1956
when stations began to be set up in Antarctica as part of the International
Geophysical Year (IGY) of 1957 - 58.
Observing stations using the Dobson ozone spectrophotometer were set up
at Argentine Islands (which later became Faraday and is now Vernadsky) and at
Halley and these have continued without interruption. In the 1960s further measurements were begun at Amundsen-Scott,
Base King Baudouin, Byrd, Hallett and Syowa, though only the first and last of
these continue today. In the 1970s
ozone measurements from polar orbiting satellites began, and a variety of
satellites continue to make measurements.
A few stations have made ozone profile measurements using chemical
sondes and this is also possible from ground based measurements using the
Umkehr technique.
The long record from Halley and its location
within the winter polar vortex made it the best site for discovering what is
now known as the Antarctic ozone hole [Farman, Gardiner & Shanklin,
1985]. The seasonal pattern of the
total ozone column in Antarctica is linked to the development and breakdown of
the winter circumpolar vortex.
Historically ozone values were around 300 DU at the beginning of the winter (March in Antarctica), and similar at the end. During the winter, ozone amounts build up in a circumpolar belt just outside the vortex, due to transport of ozone from source regions in the tropics. Very high values, occasionally exceeding 500 DU may be seen in this belt, which typically lies between about 60° S and 40° S. When the polar vortex breaks down in the spring (October), this belt of high ozone column air sweeps across Antarctica, typically reaching the area around the central Pacific sector first and the Atlantic sector last. Peak values used to exceed 450 DU, and then slowly declined back to 300 DU during the summer and autumn.
Since the mid 1970s an increasingly different pattern of behaviour is seen – the Antarctic Ozone Hole. At the end of the winter, values are around 10% lower than they were in the 1970s and drop at around 1% per day to reach around 100 DU at the end of September. Values then slowly begin to recover, but the spring warming in the stratosphere is often delayed to the end of November or into December. Peak values in the spring warming are now substantially lower than in pre-ozone hole times. Figure 4 shows the seasonal variation of total ozone at Halley for the period 1957 to 1999 and the decline in late spring and early summer values since about the early seventies is very evident as is the presence of very low October values since about the 1980s.
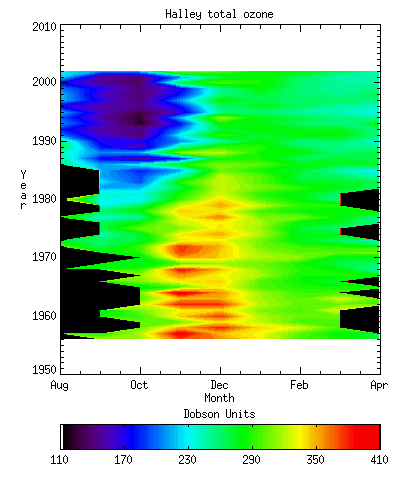
Figure 4 - The variation in
ozone at Halley from 1956 to 2002. The
ozone hole first appears in the late 1970s.
[British Antarctic Survey]
Ozone profiles can be
measured using chemical sondes, which record the ozone concentration as the
balloon rises through the atmosphere.
The most frequent technique is to bubble air through a potassium iodide
concentration cell, which generates a current that is proportional to the ozone
concentration. These sonde profiles
show that ozone concentration is low in the troposphere, but normally rises
from the tropopause to reach a peak at around 17 to 22 km, thereafter
falling. During the spring depletion,
the majority of the loss occurs between 12 and 20 km altitude and virtually all
ozone within this layer is destroyed.
[Figure 2] Although the peak
ozone concentration is around 20 km altitude the maximum absorption of short
wave solar radiation takes place at a much higher altitude of around 50
km. This absorption gives rise to the
temperature maximum in the upper stratosphere.
The mechanism which controls the development of the Antarctic ozone hole is linked to the dynamics of the winter polar vortex. During the winter, lower stratospheric temperatures drop below –80° C and at these temperatures stratospheric clouds can form. (Figure 5). Observers at stations along the Antarctic Peninsula regularly see these clouds during the late winter as Nacreous or Mother of Pearl clouds, created by lee-waves off the mountains of the Peninsula. More southerly stations sometimes report them as ‘ultra-cirrus’, which may cover the entire sky in a faint milky veil. Occasionally there are reports of clouds which resemble noctilucent clouds and it is just possible that such mesospheric clouds are seen in the Antarctic winter; these reports need further investigation. Once the clouds have formed, chemical reactions take place on the cloud surfaces, which together lock up nitrogen oxides and water and liberate chlorine and bromine (from CFCs, Halons and other similar chemicals) in an active form. When exposed to sunlight, catalytic reactions take place, which rapidly destroy ozone.

Figure 5 – Nacreous clouds
over the Antarctic Peninsula [British Antarctic Survey]
The simplest catalytic cycle that destroys ozone involves chlorine with chlorine monoxide (ClO) as an intermediary:
Cl + O3 -> ClO + O2
ClO + O -> Cl + O2
The same amount of chlorine is present at the beginning and end of the reaction and is the catalyst for the reaction to take place. Bromine can take part in similar cycles. More complex reactions also take place and together they can destroy ozone at around 1% a day until the catalytic cycle is broken, usually by other species reacting with the chlorine or chlorine monoxide to convert it into an inactive form. Paul Crutzen, Mario Molina and Sherwood Rowland carried out pioneering work on these catalytic cycles in the 1970s and in 1995 they received the Nobel prize for their work.
Satellite images give a
global picture and show the formation of the ozone hole within the strong
winter polar stratospheric vortex.
Globally ozone amounts are generally lowest at the Equator and highest
at sub-polar latitudes. The polar
vortex acts as a barrier and hence ozone rich air builds up around it, in
sub-polar latitudes. Inside the vortex,
ozone depletion is enhanced by chemical reactions on polar stratospheric
clouds, which form in the very cold vortex. When the circumpolar vortex is at
its strongest the ozone hole tends to be roughly circular, (see, for example,
Figure 6) but as it weakens the vortex often becomes strongly elliptical
(Figure 7) and often offset from the pole towards the Atlantic. At these times
the northern edge of the vortex can reach as far north as 50° S in the Atlantic
sector, posing a risk of increased UV exposure for the inhabitants of southern
South America and the sub-Antarctic islands.
The wave rotation period of the vortex is typically around a fortnight
and this can be used to give rough forecasts of the period when such areas are
most at risk. As the vortex warms the
clouds begin to disappear and chemical depletion ceases. With further warming the vortex begins to
break down and the sub-polar ozone rich air sweeps across the continent.
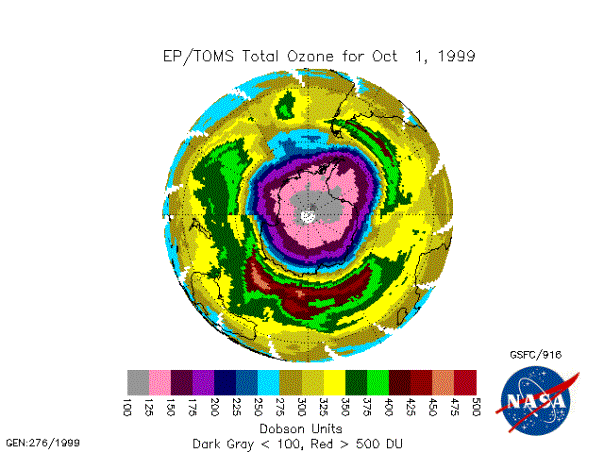
Figure 6 – Satellite image
showing the ozone hole in early October [NASA/GSFC]
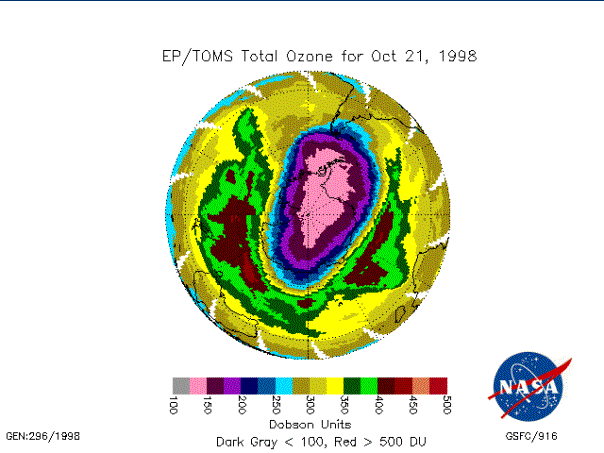
Figure 7 – Satellite image
showing the ozone hole extending towards south America [NASA/GSFC]
The lower stratospheric temperature is strongly correlated with the total column ozone at any one location, a relationship first described by Reed and Normand in 1950. The mean 100 hPa temperature over Antarctica during October to December is now significantly lower than it was 30 years ago. [Figure 8] This is due to a combination of the delayed spring warming and the lower amount of ozone in the stratosphere. Figure 9 shows the total October ozone at Halley since records began: the decrease from the mid 1970s onwards is very marked. This stratospheric change has potential implications for the surface climate, though the direct coupling between stratosphere and troposphere is weak.
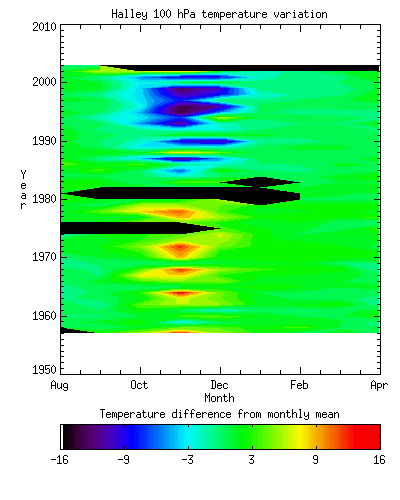
Figure 8 - The variation in
100 hPa temperature Halley at Halley from 1956 to 2003. Note the progressive spring-time
cooling. [British Antarctic Survey]
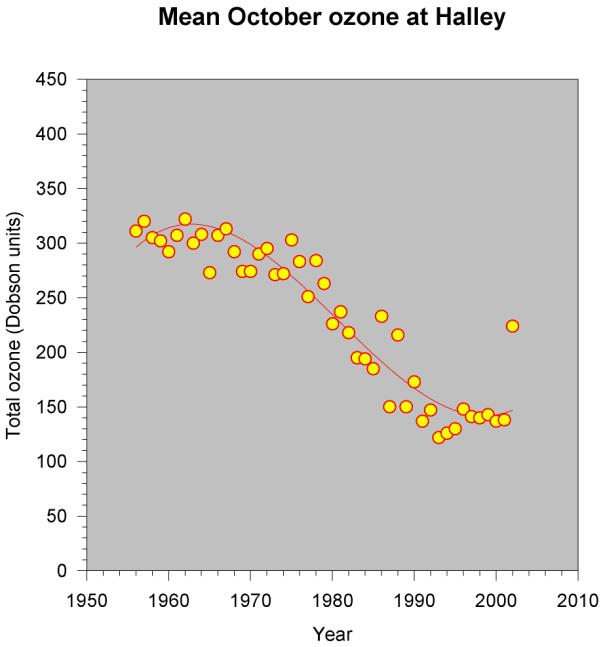
Figure 9 - The October mean
ozone at Halley 1956 - 2002. Note that
there has been little change in the mean since the late 1990s. 2002 was anomalous. [British Antarctic Survey]
The first signs of ozone depletion
can in retrospect be seen from the mid 1970s, however the change did not become
significant enough to be noticed until the early 1980s. Since then the ozone hole has grown deeper
and lasts longer. In addition, the late
spring maximum has become much reduced and there are signs of depletion in the
autumn. [Figure 4] During the late
1990s the maximum depth of the ozone hole stabilised, reflecting the nearly
complete destruction of ozone in the vulnerable part of the atmosphere.
The size and depth of the ozone hole at its maximum extent now seem to be near their peak. The 2000 and 2003 ozone holes were the largest yet seen and reached an area of 28.4 million square kilometres. The ozone hole in 2002 was anomalous as a result of unusual stratospheric conditions and does not represent a recovery. The size of the hole is constrained by the circumpolar vortex, which in turn is constrained by the size of the Antarctic continent. The depth of the hole is constrained by the height range where temperatures are cold enough for the stratospheric clouds to form. We should see a very slow return to the pre-1970 situation, provided that the Montreal Protocol is adhered to, and that there are no other changes to the atmosphere.
The Montreal Protocol is an
international agreement that is designed to reduce the total chlorine and
bromine load in the stratosphere. These
gases largely reach the stratosphere in the form of CFCs and Halons, which were
liberated in the troposphere from aerosol, refrigeration, foam blowing and fire
extinguishing systems. They have a
typical stratospheric lifetime of 50 years, hence the expectation of a return
to pre-1970 conditions on that timescale.
The protocol was first drawn up in September 1987 and subsequent reviews
have made it ever more stringent. The
original agreement was to halve the use of CFCs by 1999, with the 1990
amendment agreeing a complete ban by 2000.
The developed world achieved the target well in advance of this date and
production of other ozone depleting chemicals will stop in the early years of
this century. The developing countries
were allowed a longer timescale to cease production and some took full
advantage of this to continue production.
This has delayed the timing of the maximum halogen loading of the
atmosphere into the twenty first century.
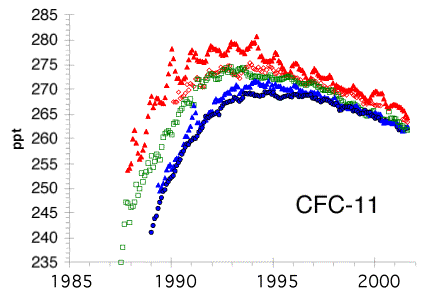
Figure 10 – Atmospheric
loading of CFC -11. A slow decline,
instigated by the Montreal Protocol, commenced in the early 1990s. [NOAA/CMDL]
Other changes are
however happening to the atmosphere, in particular the rising level of
greenhouse gases. These act to warm the
lower troposphere, but also act to cool the lower stratosphere. This may prolong the period when stratospheric
clouds can form, and hence enhance ozone depletion. This may delay the recovery of the Antarctic
ozone hole past the middle of the 21st century. It also raises the possibility of an Arctic
ozone hole forming in the next 20 years.
At present the northern polar vortex is relatively weak and usually
breaks down in January or February, much earlier in the corresponding season
than in the Antarctic. The combined
effect of slowly declining ozone throughout the Northern Hemisphere and the
increase in concentration of greenhouse gases is leading to a cooling of the
Arctic stratosphere. The colder
stratosphere and the increased temperature gradient between equator and pole
increase the strength of the circumpolar vortex making it more stable. Because it lasts longer, the spring warming
is delayed, stratospheric clouds are present into the spring and photochemistry
rapidly destroys ozone.
Other
short lived events can also affect the ozone layer. Space shuttle launches briefly perturb the stratosphere, but
their effect is soon past. Major
volcanic eruptions, such as Mt Pinatubo in the Philippines, which put several
cubic kilometres of material into the stratosphere in 1991 can affect the ozone
layer for a few years and can trigger depletion in parts of the layer which are
not normally affected by the stratospheric clouds. The greatest Antarctic depletion yet seen occurred in 1992,
however turbidity measurements show that the material had largely dissipated by
the mid 1990s. A Tunguska like event
could also damage the ozone layer.
All
living cells contain DNA, a complex molecule that carries the genetic code,
which describes the structure and biochemistry of the organism. UV can harm living things by damaging their
DNA, which readily absorbs high energy UV-B radiation. Many plants contain the pigment chlorophyll,
which absorbs visible light as the energy source for photosynthesis. It also absorbs UV-B light and becomes
bleached and non-functional. Living
things must therefore protect themselves from UV-B. Many microbes, plants and animals can synthesise protective
pigments, for example the brightly coloured lichens found encrusting rocks.
The
concerns about ozone depletion focus on the possibility of increased risk of
skin cancer. The ozone layer provides a
screen against harmful ultraviolet light, which can cause sunburn, skin cancers
and cataracts. The frequency of
occurrence of skin cancer is undoubtedly increasing, but this is more related
to a change in lifestyle since the 1960s than to ozone depletion. Our skins form brown melanin under gentle
exposure, but over exposure leads to sunburn.
There is some evidence that the cancer forming process is a two stage
one. Sunburn as a child can sensitise
the body, and subsequent exposure as an adult can trigger the cancer 20 or 30
years later. Someone deliberately going
out in the sun and lying on a beach to get a sun tan when on holiday is likely
to eventually suffer the consequences, irrespective of the protection offered
by the ozone layer.
Our
planet has an oxygen rich atmosphere with an ozone layer and together they
sustain life at the surface. Detection
of an ozone layer on a planet circling another star may be one sign that life
exists on its surface.
References and further
reading:
Chapman, S.
(1930). A theory of upper-atmosphere ozone. Mem.
R. Met. Soc., 3, pp 103-135.
Craig, R. A.
(1965). The Upper Atmosphere: Meteorology
and Physics. New York and London: Academic Press.
Farman, J. C., Gardiner, B.
G. and Shanklin, J. D. (1985). Large losses of total ozone in Antarctica reveal
seasonal ClOx/NOx interaction. Nature, 315, pp 207-210.
Gardiner, B. G. (1989). The Antarctic ozone hole. Weather, 44, pp 291-298.
Leffell, D. J. and Brash, D.
E. (1996). Sunlight and Skin Cancer. Scientific
American, 275, pp 38-43.
Schonbein, C. F.
(1840). Hebd. Scanees Acad. Sci., 10, pp 706-710.
Shanklin, J. D. (1998). The Antarctic Ozone Hole. British
Antarctic Survey.
Solomon, S. (1999).
Stratospheric Ozone Depletion: A Review of Concepts and History. Rev. Geophys., 37, pp 275-316
Stolarski, R. S. (1988). The
Antarctic Ozone Hole. Scientific American,
258, pp 20-26
Toon, O. B. and Turco, R. P.
(1991). Polar Stratospheric Clouds and Ozone Depletion. Scientific American, 264,
pp 40-47.
Scientific Assessment of
Ozone Depletion: 1998. World
Meteorological Organisation Global Ozone Research and Monitoring Project -
Report No. 44.
Stratospheric Ozone 1996.
United Kingdom Stratospheric Ozone Review Group.
The Ozone Layer. The
Department of the Environment, Transport and the Regions.
The ozone FAQ: http://www.cis.ohio-state.edu/hypertext/faq/usenet/ozone-depletion/top.html
The BAS Ozone
Page: http://www.antarctica.ac.uk/met/jds/ozone